NeuroStar Advanced Therapy (TMS) is safe and easy to tolerate. Because it is not a depression drug, NeuroStar TMS Therapy does not have the same side effects that are associated with traditional antidepressant medications. More than 1.6 million treatments have been performed with NeuroStar, further proving its safety and tolerability record.
NeuroStar TMS Therapy is free of systemic side effects associated with specific MDD treatments
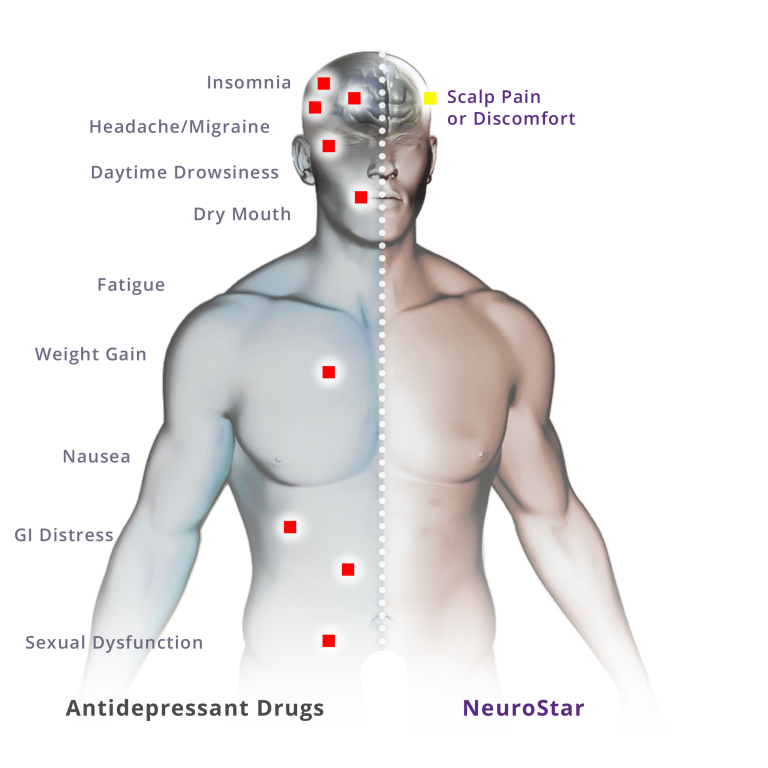
NeuroStar TMS Therapy Side Effects
In clinical trials, fewer than 5% of people discontinued treatment due to adverse events. The most common side effect was temporary pain or discomfort at or near the treatment site during therapy that usually resolves within the first week of treatment.
Other side effects (occurring in ≥5% of people and twice the rate of placebo) include eye pain, toothache, muscle twitch, facial pain, and pain of the skin.
There is a rare risk of seizure associated with TMS therapy that occurs in .01% of people. There is no adverse effect on cognition.
People should notify their doctor if they experience worsening depression symptoms, signs, or symptoms of suicidal behavior and/or unusual behavior. Family members and support individuals should also be aware of the need to observe their loved ones and notify their treatment provider if symptoms worsen.
NeuroStar Advanced Therapy (TMS) should not be used with people who have non-removable conductive metal or stimulator implants in or near the head or people who have active or inactive implants such as deep brain stimulators, cochlear implants, and vagus nerve stimulators.
Most Insurances Accepted
Fill out the form below to get in touch with us for more details and schedule an appointment
NeuroStar TMS Therapy is only available by prescription. A doctor can help decide if NeuroStar TMS Therapy is right for you.
Important Safety Information
The most common side effect is pain or discomfort at or near the treatment site. These events are transient; they occur during the TMS treatment course and do not occur for most patients after the first week of treatment. There is a rare risk of seizure associated with the use of NeuroStar TMS (<0.1% per patient). NeuroStar TMS Therapy should not be used with patients who have non-removable conductive metal in or near the head. NeuroStar TMS Therapy has not been studied in patients who have not received prior antidepressant treatment.